By P.H. Brocklehurst
Copyright © 2021 Paul H Brocklehurst
All rights reserved.
This Chapter was originally published in May 2021 as an Appendix in my book “The Perfect Stutter”[1]
A PDF version is available here
In this appendix, I discuss some recent developments in my understanding of the role that dopamine plays in enabling us to respond appropriately to the various stimuli we encounter in our lives, and I propose that phasic fluctuations in synaptic dopamine may play a key role in the moment-to-moment regulation of the availability of our speech plans for motor execution.
The relationship between dopamine receptor density and stuttering
Back in 2006, Alm’s presentation at the British Stammering Association’s annual conference in Telford had alerted me to how differences in the prevalence of stuttering in different age groups closely mirrors differences in the density of striatal dopamine receptors in those age groups; the prevalence of stuttering being highest among the age group in which the density of dopamine receptors is highest – which is between approximately two and four years of age (see figure A1).
Now, fifteen years later, we still do not know the reason for this correlation, nor do we know whether the densities of dopamine receptors in stutterers’ brains differ from those of non-stutterers of the same age.[2] All that is currently known for sure is that striatal dopamine receptor density and stuttering prevalence are closely correlated and that antipsychotic drugs which de-activate D2 dopamine receptors cause a reduction in the severity of stuttering symptoms – in a substantial proportion of adults who stutter.
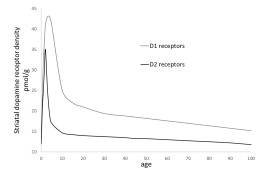
Figure A1. Showing changes in the density of D1 and D2 striatal dopamine receptors with age. Based on post-mortem data published by Seeman et al (1987), from the brains of 247 humans between 0 and 104 years of age.
Until further research is carried out, we can only speculate as to what may lie behind this correlation between striatal dopamine receptor densities and stuttering prevalence, and as there are so many details that we still don’t know, there is a risk that any such speculations may ultimately turn out to be wrong. While remaining conscious of this possibility, it seems to me that we can nevertheless glean some useful insights into possible reasons for this correlation by considering the following two questions: (1) “What function do striatal dopamine receptors normally fulfil?” and (2) “Why do young children have such high densities of them?”
The most basic answer to the first of these two questions is that our striatal dopamine receptors play a key role in enabling us to respond to stimuli – by regulating the transmission of nerve impulses from one neurone to the next. Moreover, all other things being equal, the more functional striatal dopamine receptors a person has, the more responsive to stimuli he or she is likely to be.
The two different types of dopamine receptor facilitate two different sorts of response to stimuli: D2 receptors facilitate ‘diffuse’ responses; whereas D1 receptors facilitate ‘focussed’ responses. Diffuse responses are highly variable responses – which are often referred to as ‘noise’ – reflecting the fact that the majority of such responses are unhelpful. In contrast, the focussed responses facilitated by D1 receptors are more stable and predictable – and, generally, desirable.[3] The diffuse responses facilitated by D2 receptors do, nevertheless, fulfil a vital function in enabling experimentation and learning, and it is useful to think of diffuse responses as creative, alternative responses – rather than simply as unwanted noise.
A possible clue to the answer to the second question: “Why do young children have such high densities of dopamine receptors?” can be gleaned from the finding that the density of D2 receptors peaks around two years of age whereas the density of D1 receptors peaks about a year later – around three years of age (See Figure A1). This suggests that, at two years of age, children are hard-wired to respond to stimuli in a relatively variable, creative way, whereas at three years of age they are hardwired to respond to stimuli in a more stable predictable way. This shift between two and three years of age, from a tendency to produce many variable responses to a tendency to produce a small number of stable responses, is arguably exactly what children need to achieve in order to be able to respond to stimuli in the most appropriate ways. Essentially, the initial high density of D2 receptors may enable children to produce and experiment with different responses – and thus discover which ones are most appropriate, and then the subsequent high density of D1 receptors may enable them to consolidate that learning so that they can then start to reliably respond to each stimulus with the response that they have learned is the most appropriate.
The exceptionally high densities of striatal dopamine receptors that are found in children between two and four years of age thus quite likely play an important role in enabling those children to quickly and efficiently adapt to their environment and learn how to manipulate it. Similarly, these high densities of striatal dopamine receptors are also likely to play an important role in enabling young children to develop their language and conversational skills, and they could be seen as providing a window of opportunity for intense learning, both of language itself, as well as of the social and conversational skills that go along with it.
Thus, the high densities of D2 receptors in two-year-old children quite likely lead to a tendency for their verbal responses to be highly variable and unpredictable. In contrast, around three-years-of-age, the declining density of D2 receptors, together with the high density of D1 receptors increasingly favours the execution of only the most highly activated speech motor plans – the net result being that three-year-old’s verbal responses are far less varied (less ‘creative’), more predictable and more reliable. This gradual transition – from producing many variable verbal responses to a stimulus to producing a single reliable response – is exactly what is needed in order to maximise a young child’s ability first of all to discover, and then to reliably select, the most appropriate speech plans for the various speaking situations in which he finds himself in everyday life.
Thus, by the time this window of opportunity comes to an end and their dopamine receptor densities start to fall, most children have successfully undergone vocabulary and grammar spurts, and will have learned how to modulate their speech production in social settings to elicit rewards and avoid punishments.
As I noted in Chapter 33, during this period of intense activity and learning, most children pass through a phase – between two-and-a-half and four years of age – during which they produce an increased number of stuttering-like dysfluencies. The fact that large-scale epidemiological studies have revealed an early-childhood incidence of stuttering in young children as high as 11% (Reilly et al., 2013) and 18% (Månsson, 2007) suggests that this period of increased dysfluency is likely to be a normal and largely benign phenomenon that occurs as their speech and language capacities are rapidly developing. And, as I argued in chapter 27, it is possible that this transient increase in the production of stuttering-like dysfluencies in young children is a side-effect of the fine-tuning of their release threshold mechanisms to the characteristics of the physical and social environments in which they habitually communicate. When the fine-tuning is complete and the release threshold mechanism is functioning appropriately, it should enable them to say words and phrases that are likely to elicit positive (rewarding) responses from listeners and prevent them from saying words and phrases that are likely to elicit negative (punishing) responses from listeners.
In the vast majority of children, this period of increased dysfluency lasts only a few weeks or months and then their speech once again becomes more fluent; the return of relatively fluent speech perhaps reflecting the fact that the fine-tuning of the release threshold has been successfully accomplished. However, if, for one reason or another, a child misses this window of opportunity, he or she may find it substantially more difficult to attain these communication skills after the density of their dopamine receptors has died back to the much lower levels that are typically found in older children and adults. Children who miss this window of opportunity may thus remain less skilled at adjusting their speech to suit the requirements of the speaking environment and may therefore tend to continue to elicit more punishments and fewer rewards compared to children who have developed these skills. As a consequence, such children are likely to be at risk of developing persistent stuttering.
Phasic fluctuations in dopamine release – a cause of moments of stuttering?
The early peaks in dopamine receptor densities that Alm highlighted in his thesis and associated publications (Alm, 2004; 2005) may well play an important role in providing a window of opportunity for rapid language learning and for the fine-tuning of the variable release threshold mechanism[4] to be accomplished. However, because changes in receptor density happen slowly, over a period of months, they cannot explain the fluctuations in fluency that occur from moment to moment while speaking. These moment-to-moment fluctuations may, however, potentially be successfully explained as resulting from moment-to-moment ‘phasic’ fluctuations in the quantity of dopamine released from dopaminergic neurones (the cells in which dopamine is synthesised and stored) in response to the various punishing and rewarding listener-responses that speech elicits.[5]
To clarify how such fluctuations in dopamine release may impact upon the variable release-threshold mechanism and thus also on our ability to initiate the motor execution of the words we want to say, I need, first of all, to briefly outline some findings from research that has investigated the role that dopamine plays in regulating our responses to rewards and punishments.
The role of dopamine in reward based learning
In common with all neurotransmitters, Dopamine enables nerve impulses to travel from one nerve cell to another. In particular, it facilitates the transmission of the nerve impulses that enable us to move our muscles, to speak, and also to think.
In the early 1970s, thanks to advances in electrophysiology, researchers found a way to insert micro-electrodes into dopaminergic neurones. From that time onwards, it has been possible to record the exact moments when these neurones fire and release some of the dopamine they store. Over the decades that followed, a large number of studies have been carried out on a variety of different animals (including, rats, monkeys, and more recently, human beings) that have made it possible to start to pinpoint exactly what kinds of stimuli cause these dopaminergic neurones to fire and what effect they have when they fire.[6]
As a result of these studies, the following coherent picture has gradually emerged…
In the absence of any stimuli, dopaminergic neurones will fire periodically at a low rate, releasing small amounts of dopamine and thus maintaining a background ‘tonic’ level of dopamine in the synaptic spaces that connect them to neurones further downstream. This tonic release of dopamine is responsible, amongst other things, for maintaining the background muscle tone that our muscles exhibit when in a resting state.
If an animal is then presented with a stimulus that is intrinsically rewarding (a ‘primary rewarding stimulus’), such as food or access to a sexual partner, its dopaminergic neurones suddenly start to fire at a much faster rate, releasing, within milliseconds, much more dopamine into the synaptic spaces.
This resulting increase in synaptic dopamine then stimulates dopamine receptors on downstream (post-synaptic) neurones, some of which facilitate the execution of motor plans for ‘approach behaviours’ – i.e., for muscle movements and physiological responses that enable the animal to approach (and potentially benefit from) the rewarding stimulus. In human beings, these phasic spikes in synaptic dopamine may also facilitate the execution of speech plans for the muscle movements that result in the production of words and phrases that help the person attain that rewarding stimulus or goal.
When animals and children are very young, most of the stimuli they encounter are neutral – i.e., neither rewarding nor punishing. Over time, however, neutral stimuli that consistently occur together with (or immediately prior to) a primary rewarding stimulus start to become associated with that stimulus and start to elicit similar phasic spikes in dopamine release. In this way, stimuli that were originally neutral become ‘secondary rewarding stimuli’.[7]
The transformation of neutral stimuli into secondary rewarding stimuli in this way has been confirmed in many animals, and experimental research suggests that virtually any neutral stimulus can become a secondary rewarding stimulus if it is repeatedly presented at the same time as or immediately before a primary rewarding stimulus.[8] The development of our ability to consciously or unconsciously anticipate when a primary reward is about to occur is thanks to the development of secondary rewarding stimuli in this way. Essentially, the secondary rewarding stimulus triggers the anticipation of the primary rewarding stimulus that it is associated with. Thus, for example, for Pavlov’s dogs, the sound of the bell started to trigger the (conscious or unconscious) anticipation of the food they were about to receive.
Conversely, any neutral stimulus that becomes associated with a primary punishing stimulus or with a reduction in an animal’s access to a primary rewarding stimulus will acquire the status of a secondary punishing stimulus. Secondary punishing stimuli trigger the anticipation that a primary punishment is about to occur.
Both primary and secondary rewarding stimuli have been shown to cause phasic spikes in dopamine release and to trigger approach behaviours. One might therefore expect that primary and secondary punishing stimuli would do the opposite, i.e., they would cause phasic troughs in dopamine release – which would inhibit approach behaviours. So, researchers in the 1980s were surprised to discover that, although some punishing stimuli do indeed cause phasic decreases in dopamine release, many punishing stimuli in fact seem to cause phasic increases in dopamine release, just like rewards do! For several years this unexpected finding cast doubt on the theory that the phasic release of dopamine by dopaminergic neurones plays a key role in enabling animals to respond appropriately to rewards and punishments.
In more recent years, the improved ability of researchers to track changes in the concentration of synaptic dopamine over very small time-periods led the Cambridge-based researcher, Wolfram Schultz, to propose a plausible explanation for this unexpected finding: Schultz (2016) proposed that all novel and unexpected stimuli (both rewards and punishments) lead to an initial phasic increase in synaptic dopamine which occurs before the animal has had time to identify and evaluate them. Thus, initially, all such stimuli cause approach behaviours. Then, if the animal subsequently evaluates the stimulus as ‘punishing’, the initial phasic increase in synaptic dopamine is reversed, and the animal’s synaptic dopamine levels do indeed fall – causing a phasic trough in synaptic dopamine and inhibiting approach behaviour from that moment onwards. The reason for researchers’ confusion about this back in the 1980s was because sometimes it takes a while for an animal to evaluate a stimulus as punishing, so the fall in dopamine that those researchers had expected to see in response to a punishing stimulus did not always occur straight away.[9]
Schultz (2016) proposed that the initial spike in synaptic dopamine that all novel or unexpected stimuli cause probably serves the purpose of attracting animals’ attention to those stimuli – to facilitate their ability to evaluate them. This tendency of novel or unexpected stimuli to attract an animal’s attention in this way has been well documented over many years and is known as the ‘orienting response’. Thus it seems that the initial phasic spikes in synaptic dopamine that occur in response to novel stimuli enable this orienting response to occur.
To summarise, phasic changes in the concentration of synaptic dopamine occur as follows…
- Novel or unexpected stimuli cause an initial phasic spike in synaptic dopamine levels – enabling the animal to orientate his attention towards those stimuli in order to identify and evaluate them.
- If a novel stimulus is then evaluated as rewarding, this spike in synaptic dopamine will be prolonged and its magnitude may further increase – enabling further approach behaviour towards that stimulus.[10]
- In contrast, if a novel stimulus is evaluated as punishing, the initial spike in synaptic dopamine will be reversed and a trough in synaptic dopamine levels will ensue – which inhibits approach behaviours toward that stimulus.
- Any stimulus that leads to the anticipation of a primary reward will also cause a phasic spike in synaptic dopamine, facilitating approach behaviour towards that anticipated reward.
- Any stimulus that leads to the anticipation of a primary punishment will cause a phasic trough in synaptic dopamine, inhibiting approach behaviour towards that anticipated punishment.[11]
So, how might these phasic fluctuations in our synaptic dopamine levels relate to the Variable Release Threshold Hypothesis of stuttering?
To answer this question, it is necessary to bear in mind that humans are essentially social animals, and throughout their evolutionary past, the ability to be accepted in a social group has been vital for their survival. And, to a large extent, our success in being accepted into a social group has depended on our ability to communicate successfully with other people in that social group. So, just as we are hard-wired to find food and sex rewarding, we are almost certainly also hard-wired to experience successful communication as rewarding. In other words, for human beings, successful communication constitutes a strong primary reward – just like food and sex, whereas communication failure constitutes a strong primary punishment. Similarly, any stimulus that we have learned to associate with successful communication or that causes us to anticipate that communication will be successful will constitute a powerful secondary reward; whereas any stimulus we have learned to associate with communication failure or any stimulus that leads to the anticipation of communication failure will constitute a powerful secondary punishment. Thus our brains are likely not only to compute communication failure as punishing, but they are also likely to evaluate negative listener responses, speech errors, and of course stuttering itself as punishing. Indeed, any stimulus that causes us to anticipate communication failure will be computed as a punishing stimulus – which will result in an immediate phasic decrease in the amount of dopamine released from the dopaminergic neurones in parts of the brain that regulate muscle movements for speech (including, most notably, the striatum).
As soon as a speaker perceives or anticipates that his words will result in communication failure, the resultant phasic decrease in synaptic dopamine inhibits the motor execution of the speech plan for those words. The greater the decrease, the greater the extent of the inhibition. It would appear likely that this drop in synaptic dopamine that occurs in this way may constitute the mechanism behind the rise in the release threshold that is hypothesised to take place at such times (Brocklehurst, Lickley, & Corley, 2013). Both the drop in synaptic dopamine and the resultant rise in the release threshold will last as long as the experience of communication failure (or the anticipation of communication failure) lasts.
Because such phasic reductions in synaptic dopamine delay or prevent the motor execution of speech plans, they reduce the likelihood that a speaker will say things that will elicit negative responses from his listeners. They also reduce the likelihood that the speaker will make speech errors – because plans for words or syllables that are erroneous tend not to become so highly activated, and their execution is thus inhibited by a rise in the release threshold. However, a side-effect of such phasic reductions in synaptic dopamine is dysfluent speech. And, if a speaker’s past experiences have caused him to evaluate dysfluent speech negatively, those dysfluencies will themselves act as secondary punishing stimuli and will lead to a prolongation of the drop in synaptic dopamine, thus sustaining or even prolonging the inability of the speaker to execute the problem syllables or words. [12]
The speaker may then only regain the ability to initiate execution of a problem syllable or word after the concentration of synaptic dopamine finally rises back up to a more normal level (with the consequence that the release threshold falls back down to a lower level).
How could antipsychotic drugs lead to an amelioration of stuttering symptoms?
One possible explanation is that their effect of blocking D2 dopamine receptors causes a general reduction in responsivity. And if a speaker is less responsive, he is less likely to do things or say things that elicit negative responses from his listeners. When the speaker realises that he is eliciting fewer negative responses from his listeners, the level at which his release threshold is set falls, and he finds that he can execute planned words more easily.
A related possibility, as proposed by Alm in his thesis (which I discussed in Chapter 27), is that thanks to their effect of blocking D2 receptors, antipsychotic drugs may increase the ‘signal-to-noise ratio” of speech plans. As a result, the speech plans that the speaker tries to execute will tend to be more appropriate and to contain fewer errors. The net result, once again, is speech that is more likely to be received positively by listeners, thus prompting a fall in the release threshold and a corresponding increase in fluency. Because both of these explanations involve a learning process (whereby repeated experiences of speaking impress upon the speaker the fact that his speech is no longer eliciting so many negative responses from people and is no longer so error-prone) one would expect that the speaker would have to take antipsychotics for a few weeks before he would start to experience a related improvement in his fluency – which is indeed exactly what studies of the effects of antipsychotic drugs on stuttering have tended to find.
A further alternative explanation for the ameliorative effect of antipsychotics is that, by blocking dopamine receptors, antipsychotics dampen our sensitivity to phasic rises and falls in synaptic dopamine. So, essentially the rises in synaptic dopamine are no longer so rewarding (pleasurable) and the falls are no longer so punishing. So, although the speaker may continue to make speech errors and may continue to elicit negative responses from his listeners, these errors and negative listener responses may no longer result in such a great rise in the level at which the release threshold is set.
It occurs to me that these three explanations are not mutually exclusive and all three could contribute to an amelioration of stuttering symptoms in a person who stutters.
Salience and evaluation: Two-components of the dopamine response
In addition to providing a parsimonious explanation for why stutterers tend to stutter most when they anticipate or experience communication failure, the theoretical article by Schultz (2016), which I discussed in the sub-section on reward based learning, also provides a potential explanation for why the experience of novelty frequently results in a reduction in stuttering.
In his 2016 article, Schultz discussed the problematic finding from dopamine research that punishing stimuli often cause an initial phasic rise in synaptic dopamine levels. As I mentioned earlier, this finding eventually led to the suggestion that, rather than being associated with the perception of a rewarding stimulus, phasic rises in synaptic dopamine may occur in response to the detection of any new stimulus – positive or negative; the rise essentially signalling the detection of something novel rather than something rewarding.
To account for these findings, Schultz proposed that the dopamine response to novel stimuli may in fact be made up of two components: an initial ‘detection’ component and a later ‘evaluation’ component. Thus the initial rise in synaptic dopamine that occurs in response to all sorts of new stimuli probably fulfils the function of attracting attention to the new stimulus (the orienting response). Then, that rise is either sustained or discontinued depending on how the new stimulus is evaluated. Schultz pointed out that this initial rise makes perfect sense because, although many novel stimuli or experiences have the potential to be rewarding, the only way we can find out whether or not they really are rewarding is by first of all paying attention to them. So, it makes sense that humans (and other animals) have evolved to be attracted towards novelty and to find it (initially) attractive and even pleasurable. This is especially true of new experiences that are salient, either due to their occurring in close proximity to other rewarding stimuli, or due to their unusualness, or simply due to their intensity. (See Schultz, 2016 pp.185-187)
Thus, initially, novel experiences are generally attractive and elicit approach behaviour – when we first begin processing them. Then, once we have had time to evaluate them, they may continue to elicit approach behaviour, or they may become neutral, or they may inhibit approach behaviour.
It seems likely that the same principle must apply in novel speaking situations. And this may explain why, for people who stutter, novel speaking situations or novel ways of speaking (such as speaking with an unusual accent, with unusual speed, unusual pitch, or unusual loudness) often lead to an initial increase in fluency. Presumably, novel speaking situations and novel ways of speaking lead to an initial phasic rise in synaptic dopamine and an associated lowering of the release threshold. However, this initial fluency-enhancing effect of novelty is often short lived and may die away as soon as the speaker starts to evaluate the feedback he receives – including the responses of his audience. This initial rise in dopamine may also account for the placebo effect that often occurs when a stutterer begins a novel course of therapy for his stutter, or when he first starts taking a particular type of medication. Similarly, it could explain the transient improvement in my fluency that I experienced from hypnotherapy.
The role of dopamine in drug-induced psychosis.
Schultz dedicated a short section of his 2016 paper to a potential explanation for the phenomenon of drug-induced psychosis. In it, he explained that any drugs that cause a prolonged rise in synaptic dopamine levels may cause an abnormal prolongation of the initial ‘detection component’ of the dopamine response. This prolongation of the detection component causes it to impinge upon the ‘evaluation component’. As a consequence, a person on a stimulant drug is likely to misperceive the initial rise in dopamine which is really only signalling the detection of a novel stimulus as signalling a positive evaluation of that stimulus. Hence he is likely to make positive evaluations that are inappropriate – and which may sometimes ultimately get him into trouble. Often, only when the effects of the drug start to wear off is he likely to realise that his positive evaluations were a side-effect of the drug.
After reading Schultz’ account of this two-component response to novel stimuli, I realised that it provided a neat explanation for the initial improvement and then subsequent deterioration in my speech that I experienced as a teenager when I first started experimenting with cannabis with my friends from school…
Initially cannabis made me very talkative, and very fluent. Indeed, I don’t remember stuttering at all when I first tried it. In addition to being fluent, the pleasant feeling that the drug induced in me probably played a role in causing me to perceive that the people I chatted to while under its influence were responding positively to me. It was not until some of my friends pointed out to me that my excessive talkativeness was annoying that I realised that the drug had the capacity to make me misevaluate people’s responses and to perceive them as more positive than they really were. As soon as I realised that I was misevaluating the responses of the people I was talking to, I started worrying that I was rambling and talking too much, and my speech deteriorated – to the point where I found it almost impossible to say anything at all. Then, on subsequent occasions when I took the drug, it no longer had any beneficial effect at all on my fluency. Indeed, quite the opposite.
Of course, one’s drug-induced positive misevaluations of a situation may also elicit positive responses from other people. So it is always possible that, for some individuals who stutter, cannabis and other stimulants may continue to effect a reduction in their stuttering long after the identification component of the dopamine response has ended. This may be especially likely to occur in stutterers who do not have any underlying neurological or physical impairments that cause their speech to be error-prone, such as stutterers whose problems stem primarily from unduly perfectionistic self-expectations. It may also explain why some experimental studies – including the ones by Fish & Bowling (1965), and Langova & Moravek (1964) that I discussed in Chapter 27 – have found that temporary use of stimulants elicited a lasting reduction in stuttering symptoms in a proportion of their stuttering participants.
It occurs to me that a similar mechanism – whereby the detection phase of the dopamine response is prolonged into the evaluation phase, thus leading to a falsely positive evaluation of one’s performance – may explain why, occasionally, novel and unusual forms of therapy bring about lasting reductions in stuttering: If the novelty effect of a therapy lasts long enough to enable the development of faith in one’s ability to speak without stuttering, its results may then indeed be sustained over the long-term. This may explain why faith healers and similar sorts of practitioners occasionally succeed in bringing on lasting remissions. It may also explain why some therapists, especially those with an impressive or convincing manner, apparently succeed in eliciting better results than those with a less convincing or impressive manner, regardless of what type of therapeutic approach they adopt.
It is noteworthy that even when stutterers don’t take any form of medication and don’t undergo any therapy, the severity of their stuttering nevertheless has a tendency to fluctuate, and many stutterers (myself included) have experienced how they seem to progress in their lives through repeated cycles of remission and relapse; the periods of remission and relapse often lasting for weeks, months, or even years. It is possible that some of these fluctuations may arise as a result of long-term fluctuations in the density of dopamine receptors in stutterers’ brains. Alternatively, they may reflect long-term fluctuations in the amount of dopamine synthesised in the brain or long-term fluctuations in the ability of dopaminergic neurones to release and reabsorb the dopamine they store.
Stuttering and ADHD
In his thesis, Alm (2004,2005) presented evidence strongly suggestive of the existence of a subgroup of stutterers whose stuttering began following brain damage caused by an injury or infection in early childhood. Unlike stutterers with a family history of stuttering, these individuals frequently have a history of delayed speech and language development, poor control over their attention, and some also have a history of hyperactivity. Alm noted that the behaviour of these individuals is often similar to that of people with Attention Deficit Hyperactive Disorder (ADHD) or Attention Deficit Disorder (ADD), although their symptoms are generally not severe enough to warrant formal diagnoses of these disorders. Alm noted that stutterers showing these symptoms would generally tend to have been classified by Van Riper as having a ‘Track 2’ onset of stuttering.
Because the symptoms these individuals produce overlap to some extent with those of ADD and ADHD, I was particularly interested to discover that a hypothesis exists that attributes ADHD and ADD to an under-production of dopamine and a resultant impairment of incentive learning. Specifically, in his (2018) book, Life’s rewards: Linking dopamine, incentive learning, schizophrenia and the mind, Beninger (pp. 236-244) proposed that children with ADHD have hypoactive dopamine metabolisms that are just active enough to enable them to orient towards novel stimuli but are not active enough to enable them then subsequently evaluate those novel stimuli as rewarding or non-rewarding. As a consequence of this evaluation failure, a far greater proportion of the stimuli they encounter in their everyday lives continue to be perceived as ‘novel’ and continue to attract their attention. Thus, their capacity to ignore unimportant stimuli is much reduced and their attention continues to be orientated towards every little stimulus they encounter in their environment.
Beninger also pointed out that the hyperactivity component of ADHD is only highly prominent in very young children with the disorder; it generally diminishes as they grow older, and often disappears entirely – resulting in the lesser diagnosis of Attention Deficit Disorder (ADD). This gradual reduction in hyperactivity may reflect that fact that, as young children with ADHD grow older, they do eventually learn to recognise which stimuli are likely to be unrewarding or punishing, but because they still take longer to recognise and evaluate stimuli compared to normal children or adults of the same age, many more stimuli continue to attract their attention.
With regard to speech, Alm (2004) clarified that the subgroup of stutterers who have a history of brain injury not only tend to show signs of attention-deficits and hyperactivity, but often also produce symptoms of cluttering – including a tendency to rush ahead and leave many words incomplete. In light of Beninger’s hypothesis of ADHD, it seems likely that cluttering symptoms may occur in these children because their attention is continually being attracted towards different stimuli while they talk. The classical stuttering symptoms – of being unable to initiate speech or to move forward – may only start to occur in such children somewhat later, after repeated experiences of communication failure and eliciting negative responses from listeners.[13] Whatever the case, there is no doubt that children with ADHD do elicit a lot of negative responses from listeners, as their behaviour is often perceived as annoying – leading to the frequent perception that they are ‘naughty children’. And, even though children with ADHD may be slow to evaluate such listener feedback as negative, repeated exposure to it, together with repeated experiences of communication failure are nevertheless likely to eventually lead them to evaluate their speech as ‘not good enough’, and to anticipate that their attempts to speak will result in communication failure and negative listener responses.[14] Such negative evaluations and anticipations then would start to trigger the phasic reductions in synaptic dopamine that cause them to produce stuttering blocks in addition to their cluttering and other speech-related symptoms.
If these attention deficit disorders do indeed stem from an under-production of dopamine and/or an underactive dopamine metabolism, this would go some way to explaining why the stimulant drug Ritalin – which increases dopamine metabolism – has been found to ameliorate their symptoms – increasing the attentional control and reducing the hyperactivity of people suffering from these conditions. It also highlights the possibility that stuttering symptoms may be ameliorated in the ‘attention deficit’ subgroup of stutterers by stimulants that increase dopamine metabolism.
The role of reward-based learning in the onset of stuttering
Schultz’ two-component explanation of the initial rise in dopamine levels brought on by novel experiences also provides some potential further insights into the onset of stuttering.
Initially, young children’s experiences can be divided into three sorts – (1) those which are intrinsically pleasurable or rewarding – i.e., the primary rewards like milk and interaction with their mother; (2) experiences that are intrinsically unpleasant or punishing, like pain, isolation, and sudden loud noises; and (3) neutral experiences that are neither intrinsically rewarding nor punishing.
When very young, a large proportion of children’s experiences are neutral inasmuch as they have not yet become associated with positive or negative evaluations. As time passes, these neutral experiences then gradually become associated with such evaluations, but before these associations develop, if Schultz’ two-component theory is accurate, young children’s novel neutral experiences will initially elicit phasic rises in their synaptic dopamine levels and thus will tend to elicit orienting and approach behaviour and will tend to be perceived as pleasurable. Similarly, children’s early vocalisations are likely to elicit phasic rises in synaptic dopamine, which may partially explain why young children spend so much time vocalising and engaging in vocal play, even when there is nobody there listening to them. Essentially, they enjoy the rise in synaptic dopamine that such novel behaviour (and indeed any novel behaviour) elicits. It seems likely that the rise in synaptic dopamine that accompanies their early vocalisations keeps their release thresholds at a constantly low level, rendering it easy for them to execute whatever speech plans arise in their brains.
It is only somewhat later in their development, and after the novelty value of vocal play and other early vocalisations has worn off, that children become aware that their words can elicit both positive and negative responses from other people. And, when they do realise this, their ability to vocalise and to speak then becomes subject to the second component of the dopamine response: the evaluation component.
Once the evaluation component of the dopamine response comes into play, whenever the child anticipates that a word or phrase is likely to elicit negative listener responses his synaptic dopamine levels fall. This fall triggers a rise in the release threshold which renders it difficult or impossible for him to speak that word or phrase out loud.
Thus, Schultz’ two component dopamine response predicts that children’s initial vocalisations and first words will always be executed with ease, and that their stuttering-like dysfluencies should only start to appear at the point of time in their development when their vocalisations are no longer ‘novel’, and they start to notice that some of those vocalisations elicit negative responses from listeners. This prediction fits well with many of the observations that speech and language researchers have made in relation to the initial onset of stuttering symptoms in young children, that I discussed in Chapter 33.
Looking towards the future – The importance of stuttering subtype awareness
There is already a consensus among stuttering researchers that there are at least two major stuttering subtypes, and a recognised priority for ongoing research is to clearly identify the characteristics of these subtypes. Historically, researchers have distinguished between ‘developmental stuttering’ and ‘neurogenic stuttering’, and they have understandably tended to consider early-onset stuttering as ‘developmental’ and late (adult) onset stuttering as ‘neurogenic’. In categorising stuttering in this way, they have, however, consistently overlooked the fact that young children are at least as prone to suffering neurological damage from accidents, infections, etc., as are adults. Consequently, they have consistently overlooked the fact that much of the stuttering that occurs in young children is almost certainly neurogenic in origin, even though its early onset means that it does have a strong developmental dimension to it.
On the basis of the research Alm reviewed in his thesis, it is possible to identify two major subgroups of stutterers: One in which there is a family history of stuttering, and one in which stuttering starts following an event, such as an infection or injury, that results in neuronal damage (see Alm & Risberg, 2007).
As discussed in Chapter 27, stutterers in the first group tend to be highly social, with ‘easy temperaments’ and tend, if anything, to have somewhat above-average intelligence and precocious language skills; whereas those in the other group tend towards hyperactivity, have a poor level of control over their attention, and often show signs of ADHD and cluttering. They frequently also have a history of delayed or impaired language or speech development.
The characteristics displayed by the first group of stutterers (the high-performing group) are all characteristics that have been found to be associated with a high density of D2 dopamine receptors (Alm and Risberg, 2007, p.32). And it seems likely that their high density of D2 dopamine receptors enables them, as children, to learn quickly and to adapt easily to their physical and social environments. Alm (2004) proposed that stutterers whose onsets belonged to Van Riper’s tracks 1 and 3 could both fit into this high-performing category.[15]
In contrast, Alm’s second group (in whom stuttering began following a brain injury of some kind) probably contains many individuals who would have been classified by Van Riper as having Track 2 onsets. Depending on how early in their lives the brain injury occurred, many in this group may never have experienced a period of ‘normal’ speech. In this group, difficulty paying or maintaining appropriate attention, poor articulation skills, reactive temperaments, and hyperactivity may all contribute to their communication attempts frequently resulting in failure and eliciting negative responses from listeners.
Although these two stuttering subtypes may result from substantially different aetiologies, it seems likely that both share the same ‘final common pathway’ – inasmuch as the moments of stuttering that occur in both subtypes occur as a direct result of phasic reductions in synaptic dopamine, brought on by the perception (or anticipation) of communication failure.
Despite being theoretically possible, it may well never be practical to measure dopamine metabolism in stutterers attending speech therapy. Nevertheless, now that the role of dopamine in incentive learning is well established and understood, and now that we have a clearer awareness of the important role that dopamine plays in both the identification and evaluation stages of learning, this knowledge can potentially be used to develop speech therapy approaches uniquely suited to the different learning styles that likely characterise the two main stuttering subtypes. From the perspective of incentive- learning, one of the keys to successful speech therapy for stuttering would be the therapist’s ability to accurately identify the moments when the speaker is evaluating his performance. Having identified when those moments occur, the therapist then needs to find ways to ensure that, during those moments, the speaker’s synaptic dopamine levels remain sufficiently high to cause appropriate positive evaluations to occur. It is also possible that the temporary use of stimulants like Ritalin may be found to help young children with co-occurring stuttering and attention deficits.
It is my hope that, as time passes, systematic research into the details of the relationship between stuttering and dopamine will be carried out, and as a result, a clearer understanding of the roles that dopamine plays in the stuttering subtypes will emerge. My feeling is that, of all the avenues of research into stuttering that could be pursued, studies of dopamine metabolism and dopamine-mediated incentive learning in people who stutter have the potential to bear much fruit.
References
Alm, P. A. (2004). Stuttering and the basal ganglia circuits: a critical review of possible relations. Journal of communication disorders, 37(4), 325-369.
Alm, P. A. (2005). On the Causal Mechanisms of Stuttering. Lund University.
Alm, P. A., & Risberg, J. (2007). Stuttering in adults: The acoustic startle response, temperamental traits, and biological factors. Journal of Communication Disorders, 40(1), 1-41.
Barona-Lleo, L., & Fernandez, S. (2016). Hyperfunctional voice disorder in children with Attention Deficit Hyperactivity Disorder (ADHD). A phenotypic characteristic? Journal of Voice, 30(1), 114-119.
Beninger, R. J. (2018). Life’s rewards: Linking dopamine, incentive learning, schizophrenia and the mind. Oxford UK: Oxford University Press.
Brocklehurst, P.H. (2021). The Perfect Stutter. Google Play Books.
Brocklehurst, P. H., Lickley, R. J., & Corley, M. (2013). Revisiting Bloodstein’s Anticipatory Struggle Hypothesis from a psycholinguistic perspective: A Variable Release Threshold Hypothesis of stuttering. Journal of communication disorders, 46(3), 217-237.
Fish, C. H., & Bowling, E. (1965). Stuttering—The Effect of Treatment with D-Amphetamine and a Tranquilizing Agent, Trifluoperazine; A Preliminary Report on an Uncontrolled Study. California medicine, 103(5), 337.
Langova, J., & Moravek, M. (1964). Some results of experimental examinations among stutterers and clutterers. Folia Phoniatrica et Logopaedica, 16(4), 290-296.
Maguire, G. A., Riley, G. D., Franklin, D. L., Maguire, M. E., Nguyen, C. T., & Brojeni, P. H. (2004). Olanzapine in the treatment of developmental stuttering: a double-blind, placebo-controlled trial. Annals of Clinical Psychiatry, 16(2), 63-67.
Maguire, G. A., Yeh, C. Y., & Ito, B. S. (2012). Overview of the diagnosis and treatment of stuttering. Journal of Experimental & Clinical Medicine, 4(2), 92-97.
Månsson, H. (2007). Complexity and diversity in early childhood stuttering. Paper presented at the Fifth World Congress of Fluency Disorders, Dublin.
Reilly, S., Onslow, M., Packman, A., Cini, E., Conway, L., Ukoumunne, O., et al. (2013). Natural history of stuttering to 4 years of age: A prospective community-based study. Pediatrics, 132(3), 460-467.
Schultz, W. (2016). Dopamine reward prediction error signalling: a two-component response. Nature Reviews Neuroscience, 17(3), 183-195.
Seeman, P., Bzowej, N. H., Guan, H. C., Bergeron, C., Becker, L. E., Reynolds, G. P., et al. (1987). Human brain dopamine receptors in children and aging adults. Synapse, 1(5), 399-404.
Wu, J. C., Maguire, G., Riley, G., Lee, A., Keator, D., Tang, C., et al. (1997). Increased dopamine activity associated with stuttering. Neuroreport, 8(3), 767-770.
Yairi, E., & Ambrose, N. (2005). Early Childhood Stuttering. Austin, TX: PRO-ED, Inc.
[1] The Book “The Perfect Stutter” By Paul H Brocklehurst is available as an E-book on Google Play Books, and as a paperback on Amazon Kindle.
[2] In recent years, imaging techniques have evolved to the point where it is now possible, with the use of radioactive isotopes, to measure (in vivo) how active a person’s dopamine metabolism is and whether or not the density of his D2 receptors is abnormal.
[3] Alm (2004, pp.46-47) noted that higher densities of D2 receptors are associated with lower levels of inhibition of background noise (provided by the indirect pathway from the striatum), whereas higher densities of D1 receptors are associated with a stronger “signal” (provided by the direct pathway) – resulting in stronger activation of motor plans for desired movements.
[4] See Brocklehurst, Lickley, and Corley (2013), for a description of how this mechanism is believed to operate.
[5] Significant variations in the quantity of dopamine released from dopaminergic cells in our brains occur within a matter of milliseconds – which is a timescale that does have the potential to explain why, for example, a stutterer’s level of fluency may suddenly change from one word to the next and from one speaking-situation to the next.
[6] For a useful in-depth discussion of this research see Beninger (2018, pp.99-105).
[7] A good example of this process is the sound of the bell that Pavlov used in his experiments (that I discussed in Chapter 2). Initially, when Pavlov’s dogs first heard the bell, it would have constituted a neutral stimulus. But then over time, as it gradually became associated with the presentation of food, the sound of the bell would have come to elicit phasic spikes in dopamine release of a similar magnitude to those which originally occurred in response to the food itself. Thus, the sound of the bell would start to trigger salivation in the dogs even before any food was presented to them.
[8] Since the 1980s it has become evident that these phasic spikes in dopamine release play a key role in both the Pavlovian classical conditioning as well as in Operant (Instrumental) conditioning.
[9] This is especially likely to be the case with novel stimuli – which are likely to take substantially longer to evaluate than non-novel stimuli.
[10] This is true for both primary and secondary rewarding stimuli.
[11] In addition, Schultz noted that the non-occurrence of an anticipated reward also leads to a phasic trough in synaptic dopamine – which begins at the moment the animal becomes aware that the anticipated reward is not materialising, and the non-occurrence of an anticipated punishment leads to a phasic spike in synaptic dopamine – which begins at the moment the animal becomes aware that the anticipated punishment is not materialising.
[12] Alm (2004, p.343) made a similar suggestion: “A negative emotional experience of stuttering could be described as an event that was less rewarding than predicted, thereby reducing dopamine release and weakening the motor program for the intended speech sequence that failed. This mechanism might result in a ‘‘vicious circle,’’ where negative experiences of stuttering lead to increased stuttering, etc. On the other hand, positive emotional experiences of a functional speech pattern would tend to strengthen the automaticity of this pattern.”
[13] Surprisingly, the study by Langova and Moravek (1965) actually found that clutterers and ‘stutterer-clutterers’ symptoms actually improved on antipsychotics whereas ‘pure stutterers’ (without cluttering) symptoms tended to improve on stimulants – the opposite tendency to the one I predict here. (see Alm, 2004 for a discussion of their findings).
[14] I also came across some research (Barona-Lleo & Fernandez, 2016) that found that 86% of children with ADHD exhibited co-occurring signs and symptoms of dysphonia – which could further undermine such children’s confidence in their ability to communicate successfully.
[15] This would fit well with Yairi and Ambrose’s (2005) suggestion that many Track 3 stutterers probably had Track 1 stutters earlier on in their lives, many of which may have been short-lived and were therefore never diagnosed.